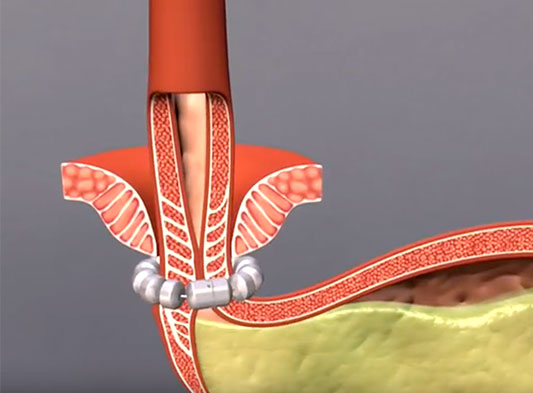
LINX is a simple procedure and mechanism. LINX is a small and flexible ring of magnets that opens with a swallow and stays closed at rest. Simple and effective.
LINX

Get off of the medication. Be free of reflux.
Risks of the Linx procedure are similar to that of a Nissen Fundoplication with the primary risks being bleeding, infection, and injury to the stomach or esophagus. Other temporary side effects include difficulty swallowing, and bloating. Risks specifically associated with the Linx device include device erosion, need for device explant, device failure and device migration.
Success rates for reduction of PPI use has been shown to be roughly 85-90% at 2 years. One study has shown 85% of patients to be free from daily dependence on medical therapy.
FREQUENTLY ASKED QUESTIONS ABOUT LINX®
HOW IS LINX® IMPLANTED?
LINX® is implanted using the minimally invasive technique of surgery known as laparoscopy. This method uses miniature incisions in the abdominal wall to place the device around the esophagus. The strengthens the weakened valve that allows reflux.
WHEN CAN I START EATING NORMALLY AGAIN?
Patients are quickly start on a normal diet. This allows the magnet to open and close soon after surgery and improve the long term effectiveness of the LINX device.®
WHEN CAN I RETURN TO NORMAL PHYSICAL ACTIVITIES?
Patients can return to normal activity within a couple of days.
WILL I BE ABLE TO BELCH OR VOMIT WITH LINX®?
Patients who have received LINX® are generally able to belch and vomit.
HOW LONG WILL LINX® LAST?
LINX® is designed to last a lifetime. The cases and wires are constructed of durable titanium, which is commonly used in medical implants such as a hip replacement . LINX® uses permanent magnets that are designed not to wear out.
CAN I GO THROUGH AIRPORT SECURITY?
LINX® is too small to set off alarms at the airport. All patients will be given ID card to have available in the unlikely event of an issue.
WILL I BE ABLE TO FEEL LINX® AFTER IT HAS BEEN IMPLANTED?
A small number of patients have reported feeling the LINX®. These sensations usually dissipate.
CAN I HAVE AN MRI?
There are two versions of the LINX device. One is safe in MRI scanning machines up to 0.7T, the other is safe to 1.5T. You should discuss the MRI scanning options with your doctor prior to deciding on treatment with LINX®.
*LINX is prescribed by your physician. It is not for everyone. Please talk to your doctor to see if it is right for you. Your physician should discuss any potential benefits and limitations with you. Although many patients benefit from LINX, results may vary. Each patient experience is unique and does not provide any indication, guide, warranty or guarantee as to the response others will have to LINX. The response other individuals have to LINX could be different. For more information about the benefits and risks of LINX please visit www.linxforlife.com or www.toraxmedical.com.
The LINX® Reflux Management System is indicated for those patients diagnosed with Gastroesophageal Reflux Disease (GERD) as defined by abnormal pH testing, and who continue to have chronic GERD symptoms despite maximum medical therapy for the treatment of reflux. Rx only.
Safety and Warnings
Contraindications: Do not implant the LINX System in patients with suspected or known allergies to titanium, stainless steel, nickel or ferrous materials.
Warnings: The LINX device is considered MR Conditional in a magnetic resonance imaging (MRI) system up to either 0.7 Tesla (0.7T) or 1.5-Tesla (1.5T), depending on the LINX model implanted. Laparoscopic placement of the LINX device is major surgery.
General Precautions: The LINX device is a long-term implant for use in patients 21 years or older. Medical management of adverse reactions may include explantation and/or replacement.
Potential Risks Associated with LINX System: dysphagia, stomach bloating, nausea, odynophagia, increased belching, decreased appetite, inability to belch or vomit, flatulence, early satiety, device erosion, device migration, infection, pain, and worsening of preoperative symptoms.

SCHEDULE TODAY!
Schedule a consultation today and free yourself from acid reflux. You have goals, we can get you there.